.svg) National Institute of General Medical Sciences |
 |
 |
National Biomedical Resource for |
| HFHF ESR: time-domain |
|
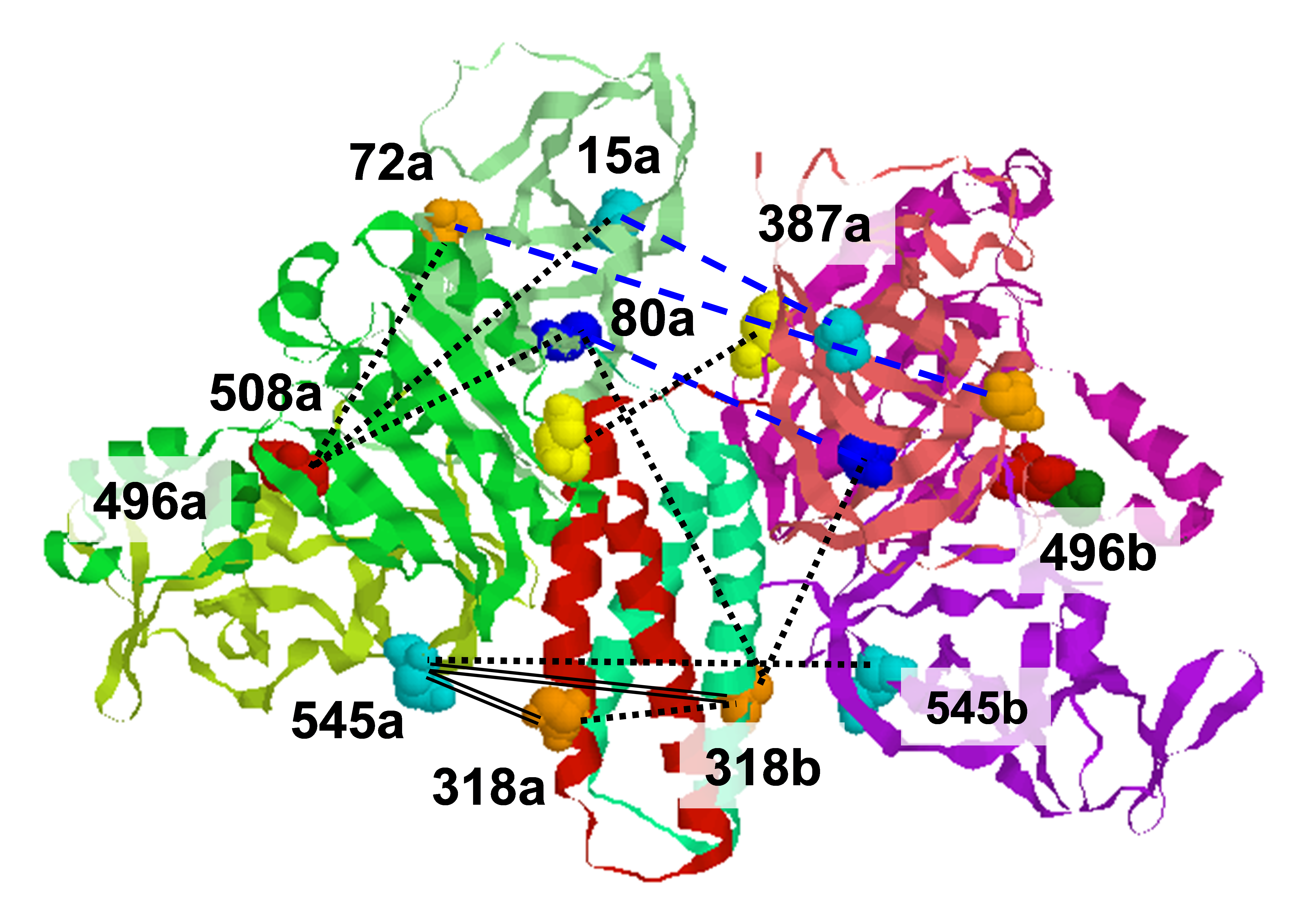
In order to understand the complex molecular dynamics of membranes and biopolymers, it is useful to have spectral information from studies at a variety of frequencies. This is because the ESR spectrum at a given frequency is most sensitive to processes that occur over a limited range of time scales. As the frequency is increased, the experiment becomes more sensitive to faster dynamical processes. We call this the ‘variable time snapshot’ feature of ESR spectroscopy. For example, ESR studies at high fields and frequencies (HFHF ESR) are particularly useful for unraveling fast internal modes in biological systems. The flexibility of time domain spectroscopy with its ability to resolve spectral features in more than one spectral dimension makes it a useful complement to continuous wave (CW) spectroscopy, which has limited resolution, confined to one spectral dimension. For example, the extra spectral dimension(s) available to time domain experiments compared to CW experiments allow one to distinguish between inhomogeneous broadening, which reports on ordering in the system, and homogeneous broadening, which reports on the dynamics. The existence and time-dependence of cross-peaks in certain classes of time-domain experiments such as 2D-ELDOR and related 2D techniques provides further information on relaxation processes that report on rotational and translation diffusion in the system. For nitroxide spin labels, HFHF ESR can also provide superior orientational resolution, analogous to the improved resolution of the chemical shift anisotropy in NMR at higher fields. This orientational sensitivity also translates into improved resolution of orientation-dependent T2 processes. Furthermore, in complex systems, components with significantly different T2’s can be separated more easily than in CW experiments. Based on our initial successes at 95 GHz in performing these sophisticated time-domain experiments, we will exploit the ‘variable time snapshot’ feature of ESR to decompose the different modes of complex motions according to their different time scales. These novel time-domain experiments have only recently become possible at high field due to the advances in instrumentation that we have accomplished at ACERT. Given that the characteristic time scales for the important dynamic modes in biological systems that we wish to study are on the order of nanoseconds to microseconds, it is necessary to generate fast, intense millimeter wave pulses in order to create the coherences that can be manipulated in the time domain to achieve multi-dimensional spectral resolution of these modes. Using our high-power pulse 95 GHz spectrometer at ACERT, we have been able to achieve 5 ns π/2 pulses routinely with 1 ns resolution of pulse separation increments. This combination of power and time resolution was essential to permit the success of, e.g., our 2D-ELDOR experiments on rapidly relaxing systems. Our successes with HFHF ESR time domain experiments rely on the use of efficient, quasi-optical, propagation techniques that combine low loss, high power-handling capability and efficient propagation of well-polarized millimeter waves. We exploit the polarization purity of our spectrometer to ‘code’ the ESR signal in an orthogonal polarization state to the high power signal generating polarization state. This makes it possible to detect the ESR time-domain signal much sooner after a pulse than would ordinarily be the case, an important consideration for rapidly-relaxing systems where long dead-times can make weak signals unobservable. Given that it is easy to scale quasi-optical techniques up in frequency, we will be able to extend our high power pulse capabilities to 140 and 220 GHz without having to radically change our design approach as resources permit. This will permit us to extend the frequency range at which distance measurements may be performed. At high fields, we expect that the orientational selectivity of HFHF ESR will allow us to measure not only distances but relative orientations of spin labels in bi-labeled proteins, leading to improved constraints for structure determinations. |
|
© 2022 |
|
About ACERT Contact Us |
Research |
Outreach |
ACERT is supported by grant 1R24GM146107 from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health. |
|||||
| ||||||||