.svg) National Institute of General Medical Sciences |
 |
 |
National Biomedical Resource for |
| ACERT's Service and Collaborative Projects | ||||
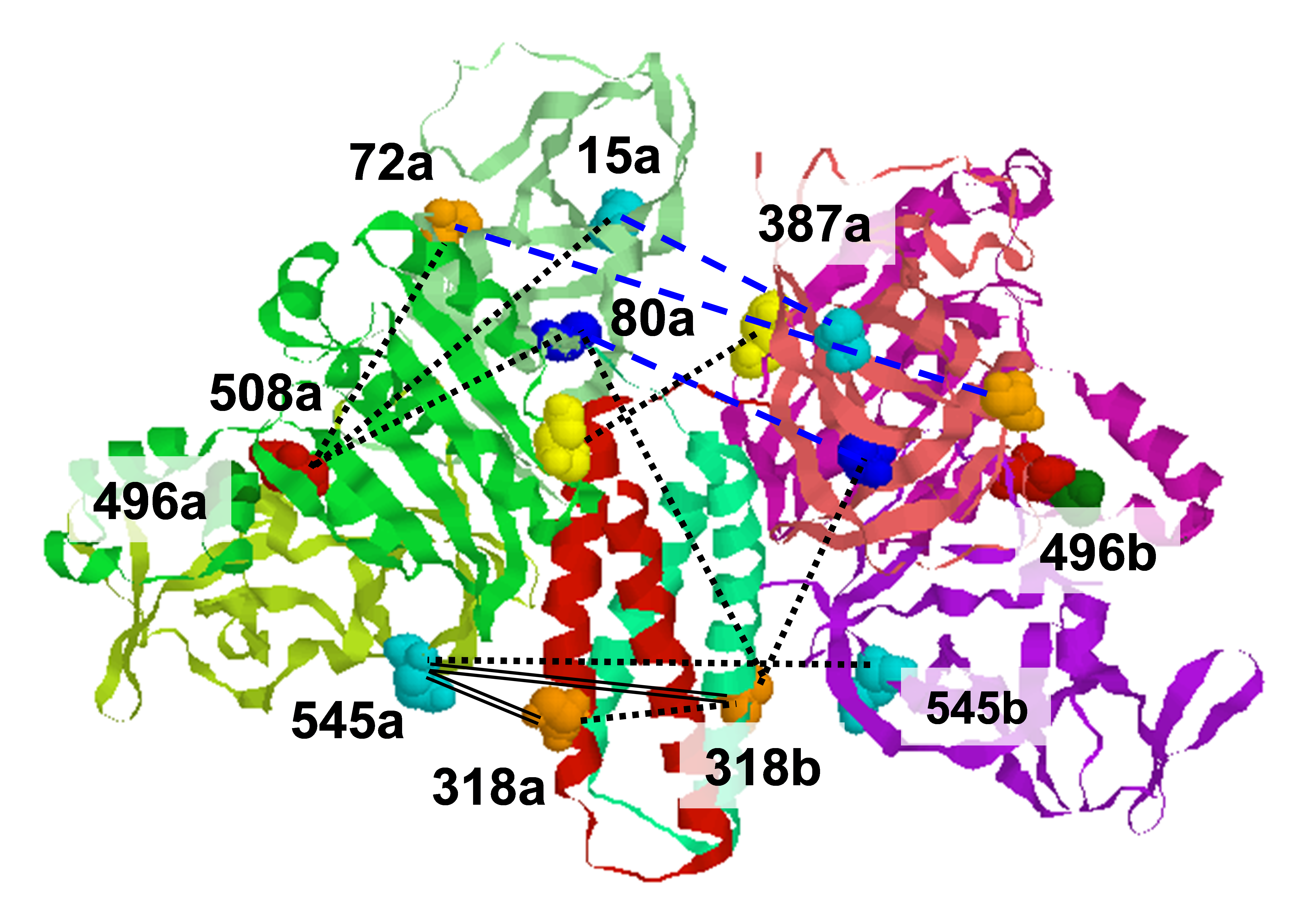
The self-assembly of the microtubule associated tau protein into fibrillar cell inclusions is linked to a number of devastating neurodegenerative disorders collectively known as tauopathies. The mechanism by which tau self-assembles into pathological entities is a matter of much debate, largely due to the lack of direct experimental insights into the earliest stages of aggregation. We made pulsed Dipolar ESR measurements of two key fibril-forming regions of tau, PHF6 and PHF6*,during the course of the aggregation. By monitoring the end-to-end distance distribution of these segments as a function of aggregation time, we showed that the PHF6(*) regions dramatically extend to distances commensurate with extended β-strand structures within the earliest stages of aggregation, well before fibril formation. Combined with simulations, our experiments show that the extended β-strand conformational state of PHF6(*) is readily populated under aggregating conditions, constituting a defining signature of aggregation-prone tau, and as such, a possible target for therapeutic interventions. Funding:R21EB022731 (to SH), NSF MCB 1158577 (to JS), P41GM103521 (to JHF). Publication: Eschmann NA, Georgieva ER, Ganguly P, Borbat PP,. Rappaport MD, Akdogan Y, Freed JH,. Shea J-E, and Han S. Sci. Rep. 7, 44739 (2017); PMCID: PMC5356194. |
||||
|
||||
|
Neil Eschmann (Department of Chemistry & Biochemistry, University of California, Santa Barbara) Elka R. Georgieva (Department of Physiology & Biophysics, Weill-Cornell Medical College, New York City) Pritam Ganguly (Department of Chemistry & Biochemistry, University of California, Santa Barbara) Peter P Borbat (ACERT) Maxime D. Rappaport, Yasar Akdogan (Department of Chemistry & Biochemistry, University of California, Santa Barbara) Jack H. Freed (ACERT) Joan-Emma Shea, and Songi Han (Department of Chemistry & Biochemistry, University of California, Santa Barbara) |
||||
|
|
About ACERT Contact Us |
Research |
Outreach |
ACERT is supported by grant 1R24GM146107 from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health. |
|||||
| ||||||||