.svg) National Institute of General Medical Sciences |
 |
 |
National Biomedical Resource for |
| ACERT's Service and Collaborative Projects | |
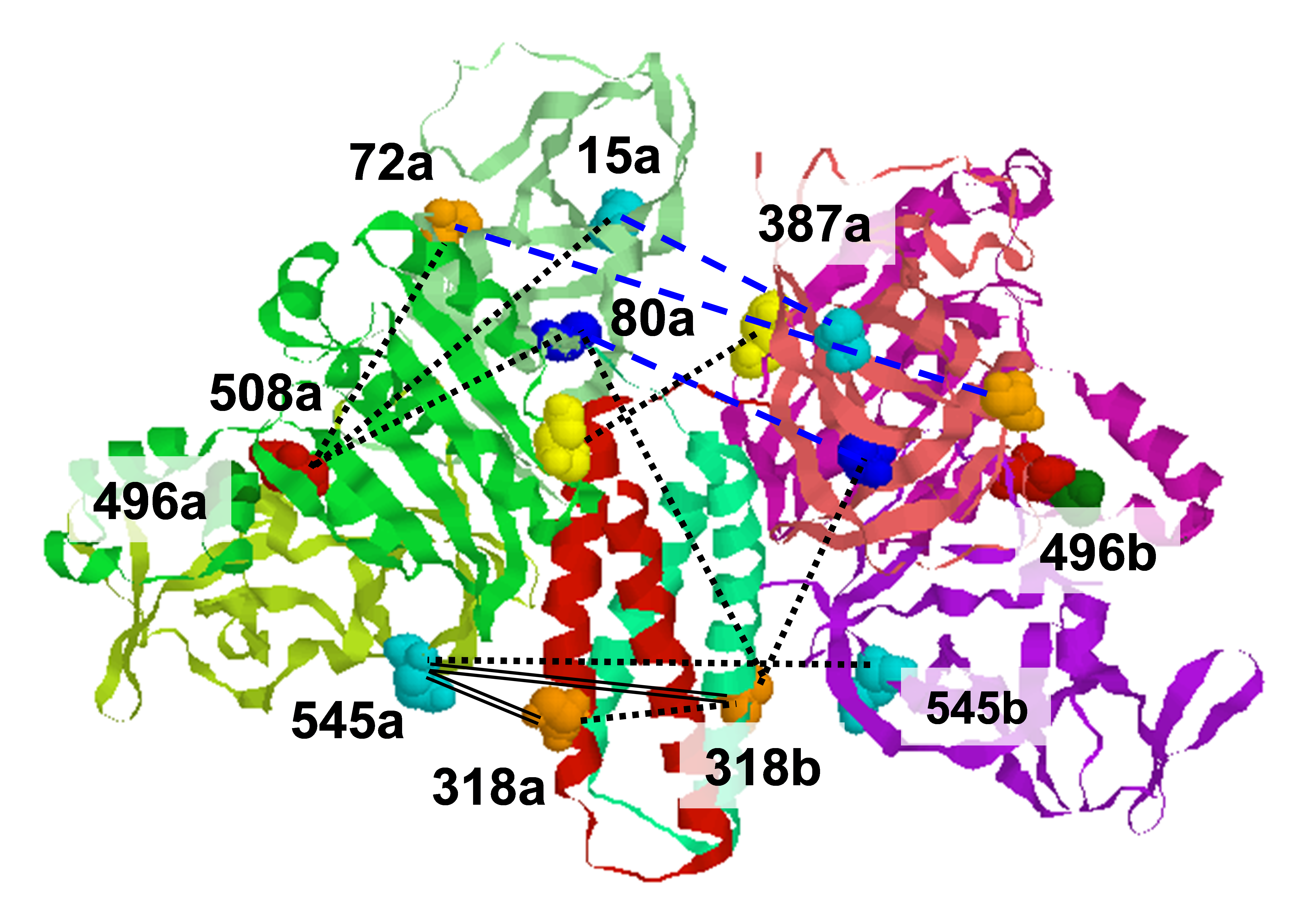
Viral fusogens merge viral and cell membranes during cell penetration. Their ectodomains drive fusion by undergoing largescale refolding, but little is known about the functionally important regions located within or near the membrane. We performed electron spin resonance studies of full-length glycoprotein B (gB), the fusogen from herpes simplex virus, complemented by the crystal structure. The membrane-proximal (MPR), transmembrane (TMD), and cytoplasmic (CTD) domains form a uniquely folded trimeric pedestal beneath the ectodomain, which balances dynamic flexibility with extensive, stabilizing membrane interactions. The postfusion conformation of the ectodomain suggests that the CTD likewise adopted the postfusion form. However, hyperfusogenic mutations, which destabilize the prefusion state of gB, target key interfaces and structural motifs that reinforce the observed CTD structure. Thus, a similar CTD structure must stabilize gB in its prefusion state. Our data suggest a model for how this dynamic, membrane-dependent 'clamp' controls the fusogenic refolding of gB. Funding: 1R21AI107171, Burroughs Wellcome Fund, Howard Hughes Medical Institute (EEH); 1F32GM115060 (RSC); P41GM103521 (JHF); R01GM123779 (JHF & ERG); in general, P41GM103403, S10RR029205, and DOE DE-AC02-06CH11357. Publication: Nat. Struct. Mol. Biol. 25, 416-424 (2018); PMCID: PMC5942590. |
|
|
|
|
Rebecca S. Cooper (Department of Molecular Biology and Microbiology, Tufts University school of Medicine, Boston) Elka R. Georgieva, Peter P. Borbat, Jack H. Freed (ACERT) and Ekaterina E. Heldwein (Department of Molecular Biology and Microbiology, Tufts University school of Medicine, Boston) |
|
|
|
About ACERT Contact Us |
Research |
Outreach |
ACERT is supported by grant 1R24GM146107 from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health. |
|||||
| ||||||||