.svg) National Institute of General Medical Sciences |
 |
 |
National Biomedical Resource for |
| ACERT's Service and Collaborative Projects | ||
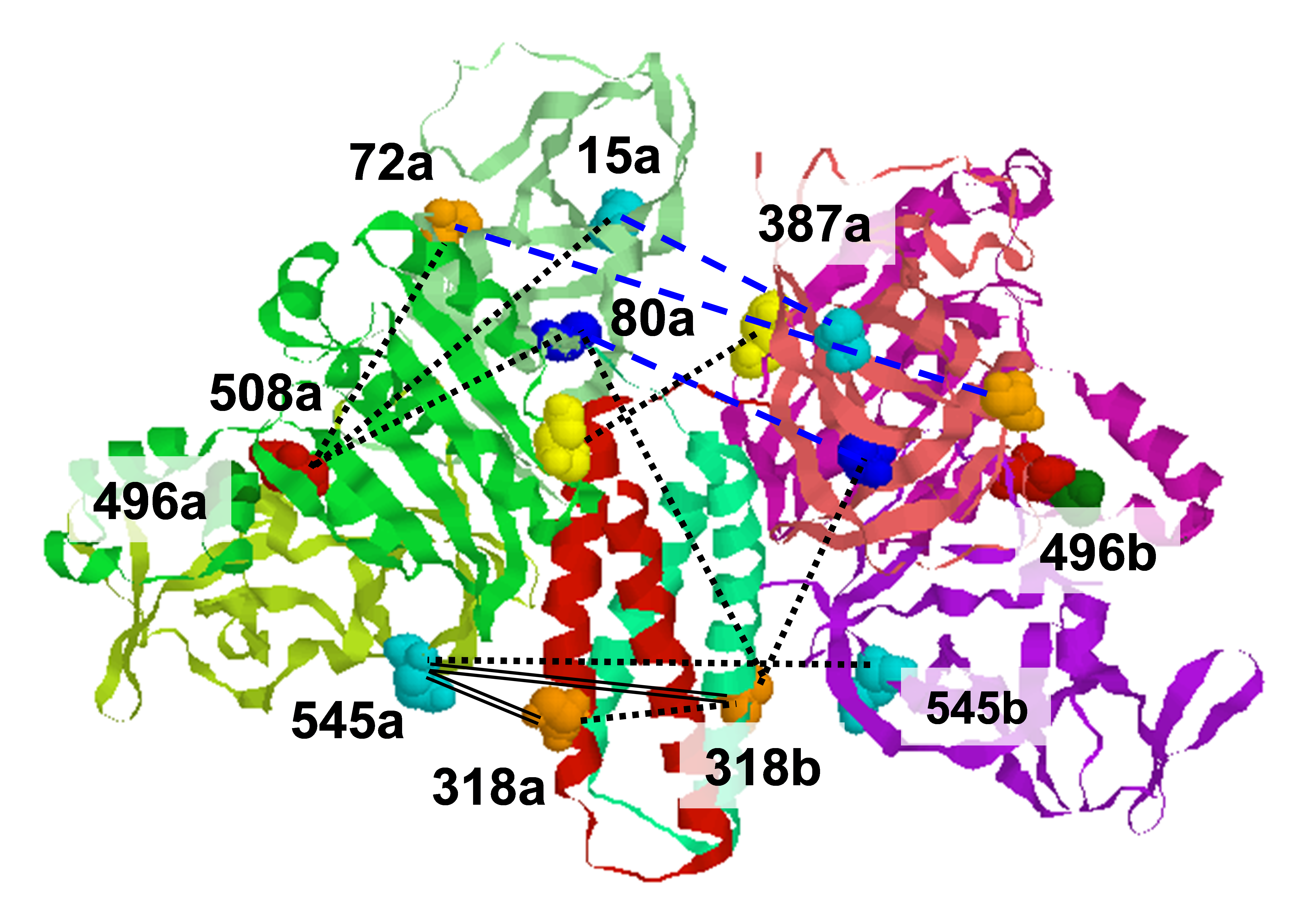
Diphthamide, the target of diphtheria toxin, is a post-translationally modified histidine residue found in archaeal and eukaryotic translation elongation factor 2 (EF2). In the first step of diphthamide biosynthesis, a [4Fe–4S] cluster-containing radical SAM enzyme, Dph1–Dph2 heterodimer in eukaryotes or Dph2 homodimer in archaea, cleaves S-adenosylmethionine and transfers the 3-amino-3-carboxypropyl group to EF2. It was demonstrated previously that for the archaeal Dph2 homodimer, only one [4Fe–4S] cluster is necessary for in vitro activity. We now demonstrate that for the eukaryotic Dph1–Dph2 heterodimer, the [4Fe–4S] cluster-binding cysteine residues in each subunit are required for diphthamide biosynthesis to occur in vivo. Our results also reveal the asymmetric functional roles of the Dph1–Dph2 heterodimer and may help to understand how the Fe–S clusters in radical SAM enzymes are reduced in biology. Funding: R01GM088276 (to HL); P41GM103521, R01GM123779 (to JHF). Publication: J. Biol. Inorg. Chem. 24, (2019), 777-782; PMC6893874. |
||
|
||
|
Min Dong (Department of Chemistry and Chemical Biology, Cornell University, Ithaca, NY, Key Laboratory of Systems Bioengineering (Ministry of Education), School of Chemical Engineering and Technology, Tianjin University, Tianjin, China) Emily E. Dando, Ilana Kotliar, Xiaoyang Su, Boris Dzikovski, Jack H. Freed (Department of Chemistry and Chemical Biology, Cornell University, Ithaca, NY) Hening Lin. (Department of Chemistry and Chemical Biology, Cornell University, Ithaca, NY, Howard Hughes Medical Institute, Cornell University, Ithaca, NY) |
||
|
|
About ACERT Contact Us |
Research |
Outreach |
ACERT is supported by grant 1R24GM146107 from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health. |
|||||
| ||||||||