.svg) National Institute of General Medical Sciences |
 |
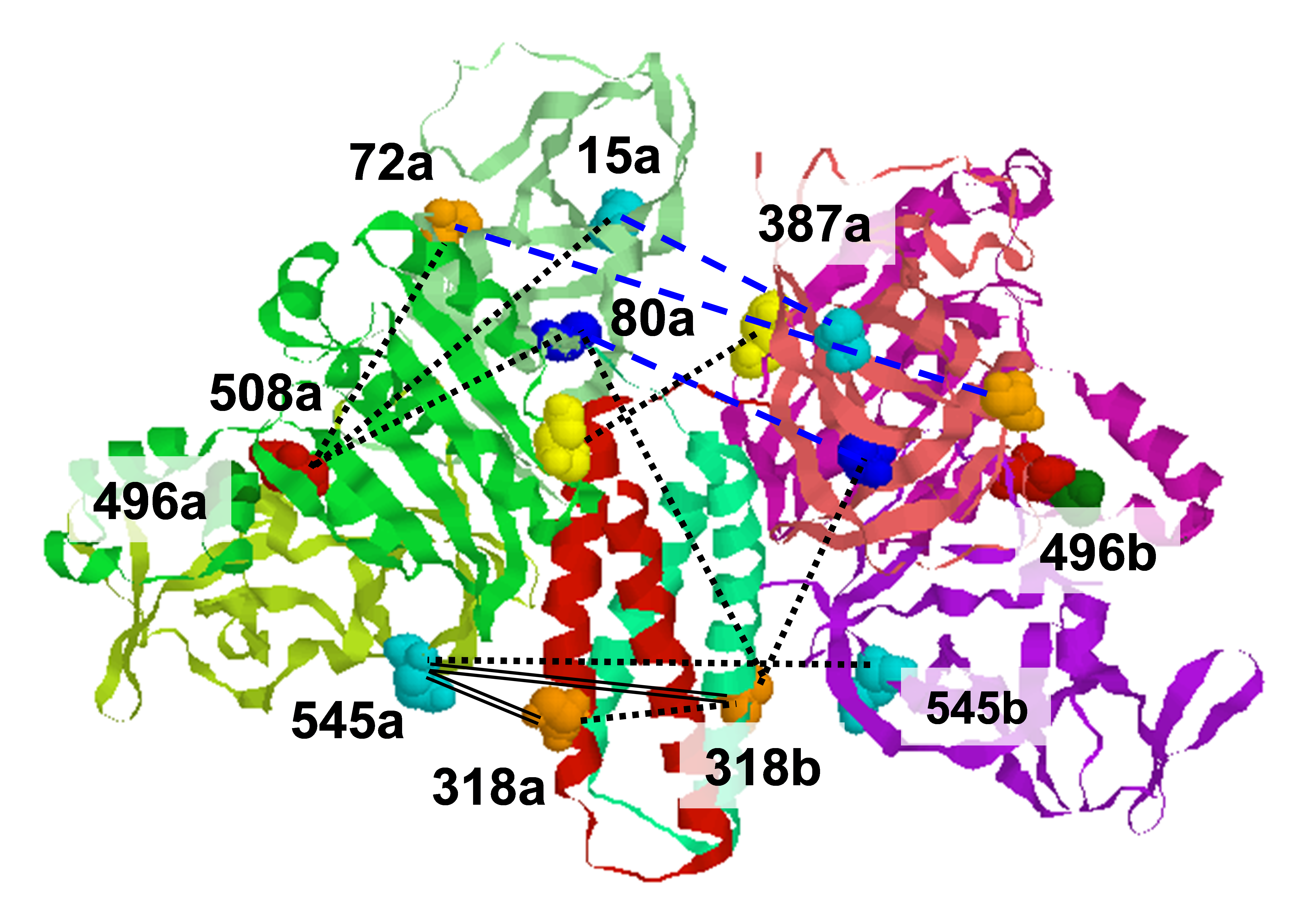 |
National Biomedical Resource for |
| MCB 0749381 (Funded by National Science Foundation / Division Of Molecular and Cellular Bioscience) | ||||||||||
| Articles: | ||||||||||
© 2022 |
||||||||||
|
About ACERT Contact Us |
Research |
Outreach |
ACERT is supported by grant 1R24GM146107 from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health. |
|||||
| ||||||||